|
7/21/2023 0 Comments Precipitate definition
The cycling of water in and out of the atmosphere is a significant aspect of the weather patterns on Earth. The water falling on land collects in rivers and lakes, soil, and porous layers of rock, and much of it flows back into the oceans, where it will once more evaporate. Definitions of PRECIPITATION A deposit on the earth of hail, mist, rain, sleet, or snow also, the quantity of water deposited. Definition (noun) a precipitated solid substance in suspension or after settling or. The water cycle describes how water evaporates from the surface of the earth, rises into the atmosphere, cools and condenses into rain or snow in clouds, and falls again to the surface as precipitation. precipitate - Dictionary definition and meaning for word precipitate. Frequent and detailed measurements help scientists make models of and determine changes in Earth’s water cycle. It's also what happens in chemical reactions when a solid settles to the bottom of a solution. Precipitation has to do with things falling down, and not just from the sky. Knowing where it rains, how much it rains and the character of the falling rain, snow or hail allows scientists to better understand precipitation’s impact on streams, rivers, surface runoff and groundwater. Precipitation is rain, snow, sleet, or hail any kind of weather condition where something's falling from the sky. So we might predict that a non-polar solvent that doesn't dissolve salts would be a bad solvent for a double replacement reaction.Precipitation is a vital component of how water moves through Earth’s water cycle, connecting the ocean, land, and atmosphere. The more you know about how the reaction occurs, and the more you know about the properties of different solvents (like their polarity), the more educated of a guess you can make! For example, in double replacement reactions, we know that the solubility of the reactants is important because we need free ions around. 
In general, it's tricky to predict for any random reaction what medium it might need. precipitation noun U (WEATHER) water that falls from the clouds toward the ground, esp. Water is a really great solvent whenever you want to have ions around. Double replacement reactions always occur in water, with the reactants in the aqueous state. Luckily, there aren't that many strong acids and bases, and you can learn morem about this from this video: Īnything that is soluble in water and dissolved (separated into individual cations and anions) is in the aqueous state. 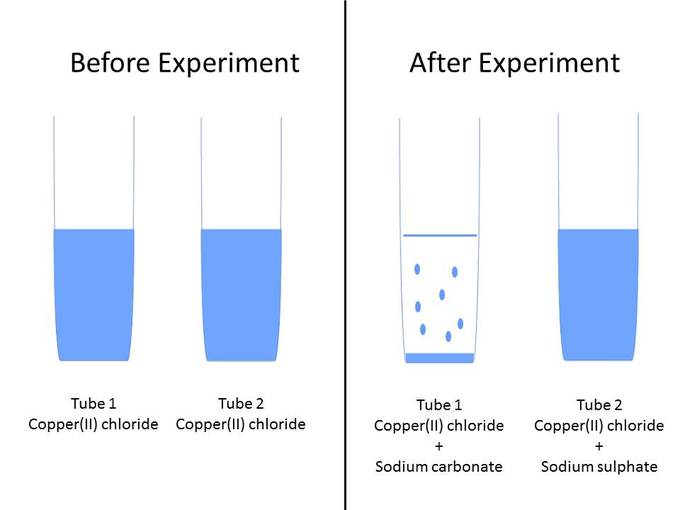
3 In case of an inorganic chemical reaction leading to precipitation, the chemical reagent causing the solid to form is called the precipitant. 1 2 The solid formed is called the precipitate. It is helpful to have the strong acids and bases memorized, since they have special reactivity. In an aqueous solution, precipitation is the process of transforming a dissolved substance into an insoluble solid from a super-saturated solution. The cation (or positively charged ion) of the salt comes from the base, and the anion (or negatively charged ion) comes from the acid. If you have tried this reaction at home, you probably remember a lot of fizzing because the neutralization reaction is accompanied by a gas-producing reaction, where the carbonic acid decomposes into carbon dioxide gas-bubbles!-and water.Ī salt is generally any ionic compound, though I have also seen it defined as an ionic compound that is formed when you react an acid and a base. A B − C D − → A D − C B − \greenD NaCH 3 COO start text, N, a, C, H, end text, start subscript, 3, end subscript, start text, C, O, O, end text.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
